Enantioselective Epoxide Opening
Abstract
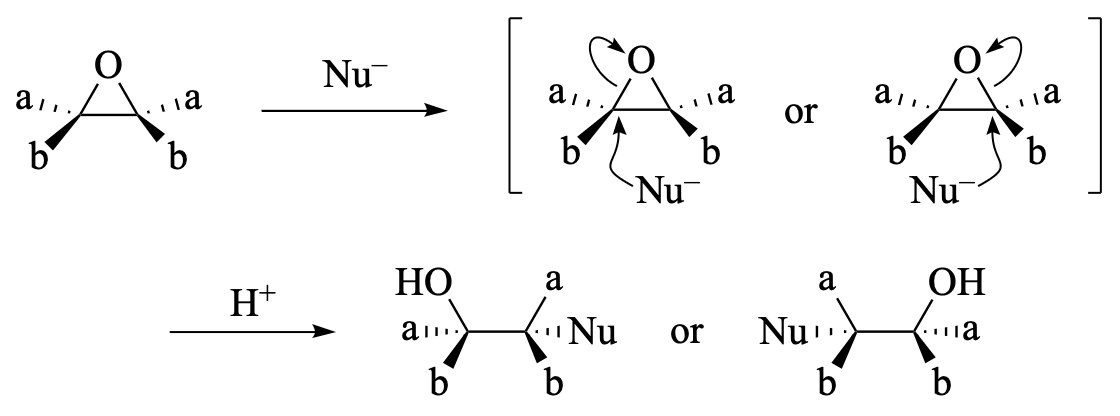
Epoxide opening is a powerful strategy for accessing β-functionalized alcohols from epoxides, which are readily available from alkenes. This review focuses on the desymmetrization of meso or centrosymmetric (prochiral) epoxides.
The enantioselective nucleophilic opening of meso epoxides with various nucleophiles (including halogen-, carbon-, nitrogen-, oxygen-, sulfur-, and selenium-centered nucleophiles) enables the efficient synthesis of enantioenriched 1,2-functionalized building blocks that bear contiguous stereogenic centers with good-to-excellent enantioselectivities. Moreover, chiral bases can accomplish the asymmetric α- or β-deprotonation of meso epoxides, thereby facilitating various unique asymmetric transformations. This review provides a comprehensive overview of these enantioselective epoxide-opening transformations using chiral reagents and catalysts, and covers the literature of enantioselective meso-epoxide opening up to April 2018. Ring-opening polymerizations, enzymatic transformations, and immobilized catalysts are not discussed, nor are kinetic and dynamic kinetic resolutions of racemic epoxides.

