A Half-Century of the Ugi Reaction: Classic Variant
Abstract
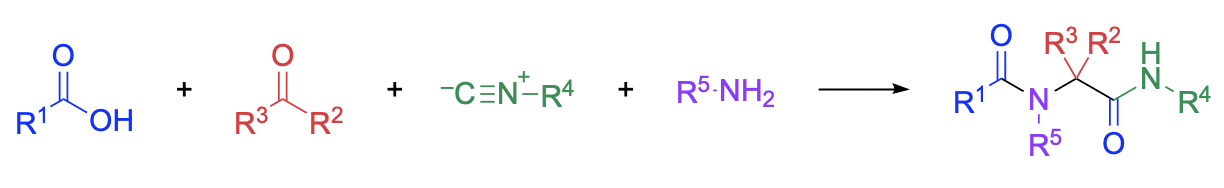 Graphical Abstract by Andrew Moore
Graphical Abstract by Andrew Moore
The classic Ugi reaction is a four-component coupling process that involves a carboxylic acid, a carbonyl compound, an amine, and an isocyanide. All reaction components are in a dynamic equilibrium with several intermediates, until an irreversible intramolecular 1,4-O→N acyl transfer leads to the formation of an N-acylamino acid amide. If chiral components are used in this reaction, a mixture of diastereomers is generally formed with moderate-to-low stereoselectivity. The broad scope of substrates that can be used in the Ugi reaction allows for the straightforward synthesis of libraries of structurally related amino acid and peptide derivatives. Therefore, this methodology is often applied to the synthesis of drugs, drug-like molecules, and natural products.
This chapter describes different variations of classic Ugi reactions, including the use of functionalized and cleavable reaction components to illustrate the broad scope of the reaction. In addition, applications in target-directed synthesis and comparisons to other related multicomponent reactions are presented. Tabular surveys are organized according to the isocyanides used. The literature is discussed from the first disclosure of an Ugi reaction in 1961 through to the end of 2012.

