A Half-Century of the Ugi Reaction: Modified Variants
Abstract
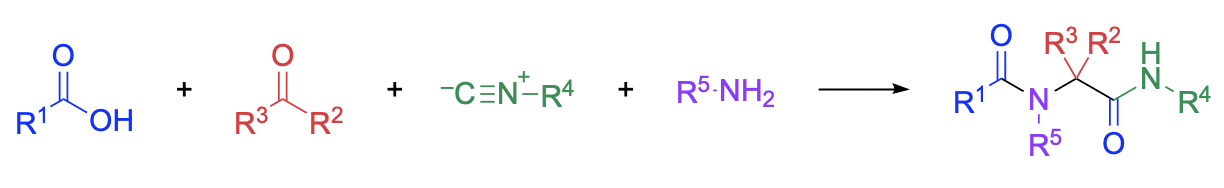
The classic Ugi reaction is a four-component coupling process involving a carboxylic acid, a carbonyl compound, an amine, and an isocyanide, resulting in an N-acylamino acid amide product. Modified Ugi reactions employ special, often multifunctional, reaction components, and can involve fewer than or more than four components. This reaction exhibits broad scope and can be used to prepare complex heterocycles. As a result, this method has been employed to prepare diverse libraries of structurally related products, and in addition, is often applied to the synthesis of drugs, drug-like molecules, and natural products. Applications in target-directed synthesis are presented, as are comparisons to other related multicomponent reactions and heterocycle syntheses; modifications leading to achiral heteroaromatic ring systems are not included. This chapter covers the literature from the early days of the Ugi reaction in 1961 through the end of 2012.

