The Cloke-Wilson Rearrangement
Abstract
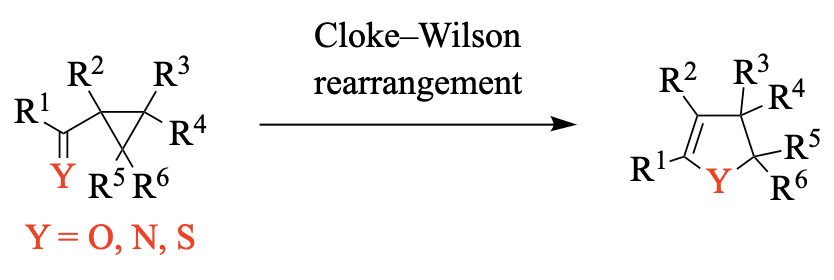
The Cloke–Wilson rearrangement is a reaction that converts cyclopropanes bearing a carbonyl, thiocarbonyl, or imino group into dihydrofurans, dihydrothiophenes, or dihydropyrroles, respectively, in a transformation that is generally driven by the release of ring strain leading to the formation of significantly less-strained five-membered heterocyclic compounds. Although the Cloke–Wilson rearrangement has typically been carried out through thermal activation of the substrate (and very often requires harsh reaction conditions), more recent research has shown that this reaction can also be carried out using Brønsted acids, Lewis acids, Lewis bases, or organometallic complexes as catalysts or promoters. These advances have contributed to broadening the functional-group tolerance of this transformation, and have allowed the Cloke–Wilson rearrangement to be used in the synthesis of densely functionalized heterocyclic scaffolds.
This chapter covers all the examples reported in the literature in which a Cloke–Wilson rearrangement has been observed, including the standard reaction that involves formyl- or acyl-substituted cyclopropanes that lead to dihydrofurans, and the heteroatom variants such as the aza- and sulfa-Cloke–Wilson rearrangements. In addition, a detailed mechanistic explanation of the different general modes of substrate activation operating with each type of catalyst/promoter has also been included, together with representative examples of practical experimental protocols and some reports in which this reaction has been applied to total synthesis.

